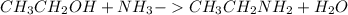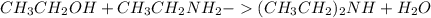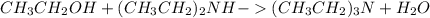Chemistry, 09.07.2019 04:30 PENTALIZARD8179
What reaction will take place if ethyl alcohol is added to a solution of hcξc: - na+ in liquid ammonia?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 23.06.2019 04:00
Silver reacts with oxygen to produce silver oxide. (write balanced chemical equation and identify type of chemical reaction.)
Answers: 1

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 10:30
What are two radioactive isotopes that are used for dating rocks that are older than 10 million years
Answers: 2
You know the right answer?
What reaction will take place if ethyl alcohol is added to a solution of hcξc: - na+ in liquid ammon...
Questions

Mathematics, 17.02.2021 14:10


Advanced Placement (AP), 17.02.2021 14:10

Mathematics, 17.02.2021 14:10

Mathematics, 17.02.2021 14:10

English, 17.02.2021 14:10

Mathematics, 17.02.2021 14:10

English, 17.02.2021 14:10

English, 17.02.2021 14:10

English, 17.02.2021 14:10


Mathematics, 17.02.2021 14:10


History, 17.02.2021 14:10

Social Studies, 17.02.2021 14:10

Mathematics, 17.02.2021 14:10

Chemistry, 17.02.2021 14:10



History, 17.02.2021 14:10