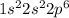Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
100 points! the electron configuration of an element is 1s22s22p6. describe what most likely happen...
Questions

Mathematics, 09.09.2020 20:01

English, 09.09.2020 20:01

Biology, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01

Mathematics, 09.09.2020 20:01


Mathematics, 09.09.2020 20:01

History, 09.09.2020 20:01



History, 09.09.2020 20:01

Social Studies, 09.09.2020 20:01

History, 09.09.2020 20:01


Mathematics, 09.09.2020 20:01