
Chemistry, 09.07.2019 17:30 1hannacarson
Consider the combustion of h2(g) 2h2(g)+o2(g)→2h2o(g). if hydrogen is burning at the rate of 0.49 mol/s, what is the rate of consumption of oxygen

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 23.06.2019 01:40
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1

Chemistry, 23.06.2019 07:30
Which of the following statements best explains why chemistry is testable a) it can measure data by experiments b) it cannot add new evidence c) it cannot be verified d) it is biased
Answers: 1
You know the right answer?
Consider the combustion of h2(g) 2h2(g)+o2(g)→2h2o(g). if hydrogen is burning at the rate of 0.49 mo...
Questions


Mathematics, 05.02.2021 22:10






Biology, 05.02.2021 22:10



Mathematics, 05.02.2021 22:10

Mathematics, 05.02.2021 22:10

English, 05.02.2021 22:10



Mathematics, 05.02.2021 22:10

Mathematics, 05.02.2021 22:10

Mathematics, 05.02.2021 22:10


Mathematics, 05.02.2021 22:10

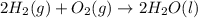
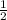 × 0.49 mol/s
× 0.49 mol/s

