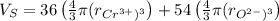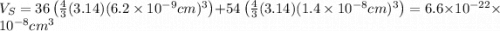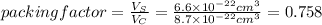Chemistry, 09.07.2019 22:00 nancye2008
The unit cell for cr2o3 has hexagonal symmetry with lattice parameters a = 0.4961 nm and c = 1.360 nm. if the density of this material is 5.22 g/cm3, calculate its atomic packing factor. the atomic weights of cr

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
When curium-242 is bombarded with an alpha particle, two products are formed, one of which is a nudge on. what is the other product
Answers: 3

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

You know the right answer?
The unit cell for cr2o3 has hexagonal symmetry with lattice parameters a = 0.4961 nm and c = 1.360 n...
Questions






Mathematics, 05.12.2020 03:00

Mathematics, 05.12.2020 03:00

Mathematics, 05.12.2020 03:00


Mathematics, 05.12.2020 03:00


Mathematics, 05.12.2020 03:00



Mathematics, 05.12.2020 03:00

Arts, 05.12.2020 03:00

Mathematics, 05.12.2020 03:00

Chemistry, 05.12.2020 03:00

History, 05.12.2020 03:00

Mathematics, 05.12.2020 03:00

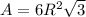
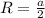
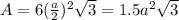
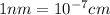
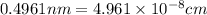
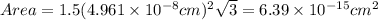
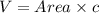
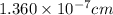
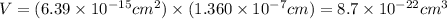
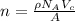
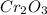 is 151.99 g/mol.
is 151.99 g/mol.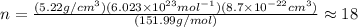
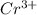 and
and 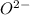 is 62 pm and 140 pm respectively.
is 62 pm and 140 pm respectively. 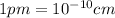
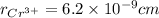
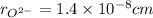
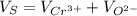
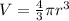 ,
,