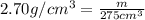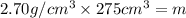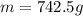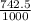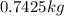Chemistry, 10.07.2019 04:00 redhot12352
Aluminum has a density of 2.70 g/cm3. if you have a volume of 275 cm3, what is the mass in kilograms

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
Aluminum has a density of 2.70 g/cm3. if you have a volume of 275 cm3, what is the mass in kilograms...
Questions







Mathematics, 25.02.2020 19:49


Mathematics, 25.02.2020 19:49








Social Studies, 25.02.2020 19:49



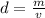
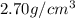 and volume i.e.
and volume i.e. 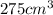 in above formula;
in above formula;