
Chemistry, 10.07.2019 06:00 iicekingmann
When silver crystallizes, it forms face-centered cubic cells. the unit cell edge length is 408.7 pm. calculate the density of silver in g/cm3?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
When silver crystallizes, it forms face-centered cubic cells. the unit cell edge length is 408.7 pm....
Questions






History, 08.10.2020 09:01



Mathematics, 08.10.2020 09:01











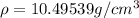

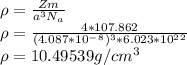
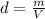
 atoms, thus, mass can be calculated as follows:
atoms, thus, mass can be calculated as follows: .
.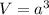
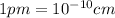
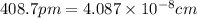
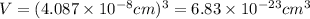
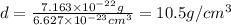
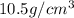 .
.

