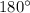Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
The expected o-c-h angle in this molecule is degrees. the expected hybridization at the central carb...
Questions



Mathematics, 28.05.2020 08:00

Chemistry, 28.05.2020 08:00

Mathematics, 28.05.2020 08:00




Mathematics, 28.05.2020 08:00

Mathematics, 28.05.2020 08:00



Mathematics, 28.05.2020 08:00




English, 28.05.2020 08:00

History, 28.05.2020 08:00

Mathematics, 28.05.2020 08:00

English, 28.05.2020 08:00

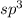 , four electron domains, as in ethane
, four electron domains, as in ethane 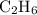
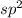 , three electron domains, as in ethene
, three electron domains, as in ethene 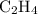
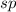 , two electron domains, as in ethyne
, two electron domains, as in ethyne 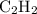
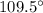 Carbon atoms with a
Carbon atoms with a 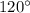 Carbon atoms with a
Carbon atoms with a