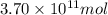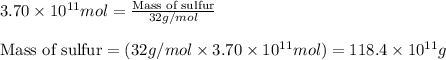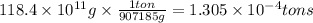Chemistry, 10.07.2019 21:00 hapjajsjjz3738
The annual production of sulfur dioxide from burning coal and fossil fuels, auto exhaust, and other sources is about 26 million tons. the equation for the reaction is s(s) + o2(g) → so2(g) how much sulfur (in tons), present in the original materials, would result in that quantity of so2?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1


Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3
You know the right answer?
The annual production of sulfur dioxide from burning coal and fossil fuels, auto exhaust, and other...
Questions

Computers and Technology, 10.10.2019 05:30



















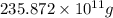 (Conversion factor:
(Conversion factor: 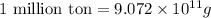 )
)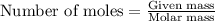 .....(1)
.....(1)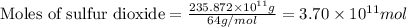
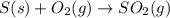
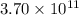 moles of sulfur dioxide will be produced by =
moles of sulfur dioxide will be produced by = 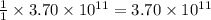 moles of sulfur.
moles of sulfur.