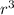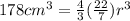Chemistry, 27.09.2019 15:00 bultron1966
You measure salt water in a tank to have a density of 1.02 g/ml. a balloon weighs 2.0 g and you weights have a mass of 30.0 g each. if you put 6 weights in your balloon, you must inflate your balloon to what diameter for it to have a density equal to the salt water, and therefor float in the middle of the tank?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 12:50
Use the standard enthalpies of formation for the reactants and products to solve for the δhrxn for the following reaction. (the δhf of c2h4 is 52.26 kj/mol, co2 is -393.509 kj/mol, and h2o is -241.818 kj.) c2h4 (g) + 3o2(g) 2co2 (g) + 2h2o(g) δhrxn = the reaction is .
Answers: 3

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3

Chemistry, 23.06.2019 12:50
Acertain reaction has a activation energy of 54.0 kj/mol. as the temperature is increased from 22c to a higher temperature, the rate constant increases by a factor of 7.00. calculate the higher temperature. c (report only numerical answer)
Answers: 3
You know the right answer?
You measure salt water in a tank to have a density of 1.02 g/ml. a balloon weighs 2.0 g and you weig...
Questions


Computers and Technology, 22.09.2019 05:10












English, 22.09.2019 05:10


Chemistry, 22.09.2019 05:10

Mathematics, 22.09.2019 05:10



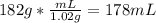
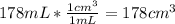
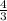 π
π