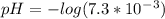Chemistry, 11.07.2019 02:30 Epicgible8136
Calculate the ph in an aqueous 0.120 m nitrous acid solution.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 21.06.2019 22:20
Much of the general structure and physical properties of the interior of the earth are inferred from: a)deep oil and gas bore holes b)geologic investigations c)analysis of seismic waves d) study of volcanoes
Answers: 1

Chemistry, 22.06.2019 00:00
The pressure in a fluid is affected by which characteristics of that fluid
Answers: 1

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1
You know the right answer?
Calculate the ph in an aqueous 0.120 m nitrous acid solution....
Questions

Biology, 02.11.2019 04:31

Mathematics, 02.11.2019 04:31

Mathematics, 02.11.2019 04:31

Biology, 02.11.2019 04:31

Social Studies, 02.11.2019 04:31


English, 02.11.2019 04:31





Business, 02.11.2019 04:31





History, 02.11.2019 04:31

Mathematics, 02.11.2019 04:31

Social Studies, 02.11.2019 04:31

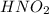 is a weak acid so first of all we solve for
is a weak acid so first of all we solve for 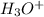 and then figure out the pH.
and then figure out the pH.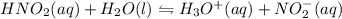
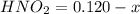

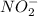 =
= 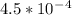 and the equilibrium expression for this would be written as:
and the equilibrium expression for this would be written as:![Ka=\frac{[H_3O^+][NO_2^-]}{HNO_2}](/tpl/images/0075/4998/f7336.png)
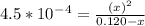
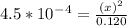
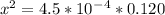
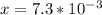
![[H_3O^+]=7.3*10^-^3M](/tpl/images/0075/4998/899ac.png)
![pH=-log[H_3O^+]](/tpl/images/0075/4998/6e71a.png)