
Chemistry, 11.07.2019 11:30 LindaCat78
Zinc metal reacts with hydrochloric acid according to the balanced equation: zn(s) + 2 hcl(aq) ¡ zncl2(aq) + h2( g) when 0.103 g of zn(s) is combined with enough hcl to make 50.0 ml of solution in a coffee-cup calorimeter, all of the zinc reacts, raising the temperature of the solution from 22.5 °c to 23.7 °c. find ∆hrxn for this reaction as written. (use 1.0 g/ml for the

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3
You know the right answer?
Zinc metal reacts with hydrochloric acid according to the balanced equation: zn(s) + 2 hcl(aq) ¡ zn...
Questions






English, 05.10.2019 00:00



Mathematics, 05.10.2019 00:00



Mathematics, 05.10.2019 00:00


English, 05.10.2019 00:00


Social Studies, 05.10.2019 00:00

History, 05.10.2019 00:00


Biology, 05.10.2019 00:00

Business, 05.10.2019 00:00

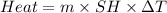
 is change in temperature.
is change in temperature.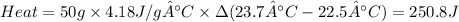
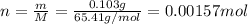
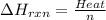
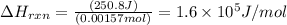
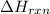 for the given reaction is
for the given reaction is 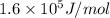
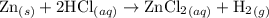
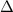 T,
T,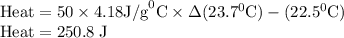
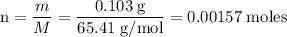
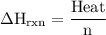
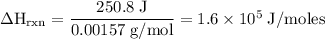
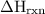 =
= 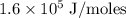 for the given equation.
for the given equation. 

