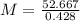The element antimony has an atomic weight of 122 and consists of two stable isotopes antimony-121 and antimony-123. the isotope antimony-121 has a mass of 121 amu and a percent natural abundance of 57.3 %. the isotope antimony-123 has a percent natural abundance of 42.8 %. what is the mass of antimony-123?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1

You know the right answer?
The element antimony has an atomic weight of 122 and consists of two stable isotopes antimony-121 an...
Questions





Health, 11.10.2019 05:00

Mathematics, 11.10.2019 05:00





Physics, 11.10.2019 05:00

Mathematics, 11.10.2019 05:00




English, 11.10.2019 05:00



Mathematics, 11.10.2019 05:00