
Chemistry, 11.07.2019 19:00 hayesvolcano
A1.42-g sample of a pure compound with formula m2so4 was dissolved in water and treated with an ex- cess of aqueous calcium chloride, resulting in the precipi- tation of all the sulfate ions as calcium sulfate. the pre- cipitate was collected, dried, and found to weigh 1.36 g. determine the atomic mass of m and identify m.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
A1.42-g sample of a pure compound with formula m2so4 was dissolved in water and treated with an ex-...
Questions



Mathematics, 27.09.2020 01:01



Mathematics, 27.09.2020 01:01


Mathematics, 27.09.2020 01:01

Mathematics, 27.09.2020 01:01


Spanish, 27.09.2020 01:01




English, 27.09.2020 01:01

Mathematics, 27.09.2020 01:01


Mathematics, 27.09.2020 01:01


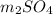 with calcium chloride can be shown as-
with calcium chloride can be shown as-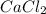 →
→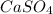 ↓+2mCl. The molecular weight of
↓+2mCl. The molecular weight of 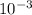 moles of calcium sulfate.
moles of calcium sulfate.

