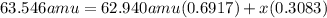Chemistry, 11.07.2019 19:30 oranjejuice
Copper has two naturally occurring isotopes and an atomic mass of 63.546 amu. cu-63 has a mass of 62.940 amu and an abundance of 69.17%. what is the identity and percent abundance of copper's other isotope? (type your answer for identity using the format cl-35 for chlorine-35.)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 23.06.2019 02:00
Scientists are often interested in knowing the molar heat of combustion – the heat released during the combustion of one mole of a substance. use the periodic table to find molar masses. how many moles of ethanol are present in the sample?
Answers: 2

Chemistry, 23.06.2019 11:20
Try to reduce the amount of leftover ingredients by changing the amount of one, two, or all three starting ingredients. show your stoichiometric calculations below. water 946.36 g sugar 196.86 g lemon juice193.37 g
Answers: 2
You know the right answer?
Copper has two naturally occurring isotopes and an atomic mass of 63.546 amu. cu-63 has a mass of 62...
Questions



Mathematics, 21.11.2019 19:31

Mathematics, 21.11.2019 19:31




History, 21.11.2019 19:31


Mathematics, 21.11.2019 19:31



Mathematics, 21.11.2019 19:31


Geography, 21.11.2019 19:31

Mathematics, 21.11.2019 19:31

Mathematics, 21.11.2019 19:31

Mathematics, 21.11.2019 19:31

Mathematics, 21.11.2019 19:31