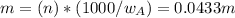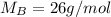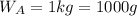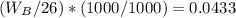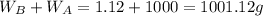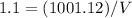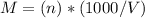Chemistry, 11.07.2019 19:30 isanaty7951
Asolution is 0.0433 m lif. what is the molarity of the solution if the density is 1.10 g/ml

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 18:30
The famous scientist galileo galilei did several experiments with sloping planes, which he rolled metal balls down so that he could study motion. by changing the slope, he could study how the speed at which the ball rolled was affected. what was the independent variable in galileo's experiment? a. the speed of the ball b. the slope of the plane c. whether the ball moved d. what the ball was made of
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

You know the right answer?
Asolution is 0.0433 m lif. what is the molarity of the solution if the density is 1.10 g/ml...
Questions

English, 05.02.2020 07:44




Mathematics, 05.02.2020 07:44

Social Studies, 05.02.2020 07:44


Mathematics, 05.02.2020 07:44


Mathematics, 05.02.2020 07:44

English, 05.02.2020 07:44

English, 05.02.2020 07:44

Mathematics, 05.02.2020 07:44



English, 05.02.2020 07:44

Chemistry, 05.02.2020 07:44

Health, 05.02.2020 07:44