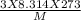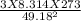Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
You know the right answer?
At 273 k and 1.00 x 10^-2 atm, the density of a gas is 1.24 x 10^-5 g/cm3. a. find vrms for the gas...
Questions



History, 26.04.2021 05:10


Physics, 26.04.2021 05:10


World Languages, 26.04.2021 05:10




Advanced Placement (AP), 26.04.2021 05:10

English, 26.04.2021 05:10


Mathematics, 26.04.2021 05:10

Chemistry, 26.04.2021 05:10


Mathematics, 26.04.2021 05:10


Mathematics, 26.04.2021 05:10

 = √
= √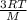 =√
=√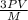 =√
=√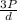 , where, R= Universal gas constant, T= Absolute temperature, P= Pressure, V= Volume of gas, d= Density of gas.
, where, R= Universal gas constant, T= Absolute temperature, P= Pressure, V= Volume of gas, d= Density of gas.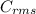 =√
=√