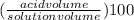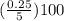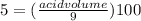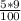Chemistry, 11.07.2019 22:30 twentyonepilots12018
Achemist has 5 l of a solution that contains 0.25 l of acetic acid. the chemist needs to prepare 9 l of a solution with the same percent concentration of acetic acid. how much acidic acid must the 9 l of the solution contain

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
Achemist has 5 l of a solution that contains 0.25 l of acetic acid. the chemist needs to prepare 9 l...
Questions




Mathematics, 24.09.2020 08:01


Social Studies, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01

Business, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01






History, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01