
(a) a 0.2 m potassium hydroxide solution is titrated with a 0.1 m nitric acid solution. (i) balanced equation: (ii)what would be observed if the solution was titrated well past the equivalence point using bromthymolblue as the indicator? (bromthymol blue is yellow in acidic solution and blue in basic solution.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 21:30
Athe top of a hill, an athlete on a skateboard has x joules of mechanical energy. how much mechanical energy will she have at the bottom of the hill? ignore the effects of friction.
Answers: 1

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1
You know the right answer?
(a) a 0.2 m potassium hydroxide solution is titrated with a 0.1 m nitric acid solution. (i) balanced...
Questions




Mathematics, 03.10.2019 02:30

Mathematics, 03.10.2019 02:30

Computers and Technology, 03.10.2019 02:30


History, 03.10.2019 02:30

Social Studies, 03.10.2019 02:30



Mathematics, 03.10.2019 02:30

Computers and Technology, 03.10.2019 02:30

Social Studies, 03.10.2019 02:30

Health, 03.10.2019 02:30

Social Studies, 03.10.2019 02:30




Social Studies, 03.10.2019 02:30

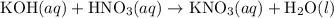
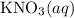 (and water) at the equivalence point. Both potassium hydroxide and nitric acid exist as strong electrolytes. As a result,
(and water) at the equivalence point. Both potassium hydroxide and nitric acid exist as strong electrolytes. As a result, 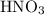 is added to the initially-basic solution. PH value of the solution would keep decreasing as the volume of the acid added increases. The final solution would be acidic as it contains not only water and
is added to the initially-basic solution. PH value of the solution would keep decreasing as the volume of the acid added increases. The final solution would be acidic as it contains not only water and 

