
Acompound contains only carbon, hydrogen, and oxygen. combustion of 9.612 mg of the compound yields 14.41 mg co2 and 3.93 mg h2o. the molar mass of the compound is 176.1 g/mol. what are the empirical and molecular formulas of the compound? (type your answer using the format co2 for co2.)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
Acompound contains only carbon, hydrogen, and oxygen. combustion of 9.612 mg of the compound yields...
Questions


Mathematics, 02.07.2021 15:50



Computers and Technology, 02.07.2021 15:50



Biology, 02.07.2021 15:50





Mathematics, 02.07.2021 15:50

Mathematics, 02.07.2021 15:50


Computers and Technology, 02.07.2021 15:50



Biology, 02.07.2021 15:50

Mathematics, 02.07.2021 15:50

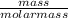 -(1)
-(1)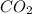 = 14.41 mg (given)
= 14.41 mg (given)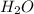 = 3.93 mg (given)
= 3.93 mg (given)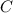 from
from 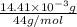
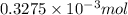
 from
from 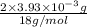
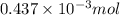
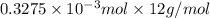
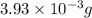
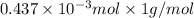
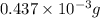
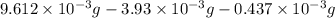
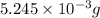
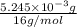
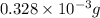
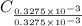
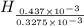
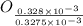
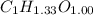
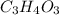
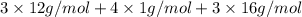
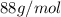
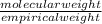 =
= 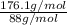
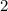
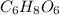 .
.

