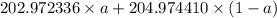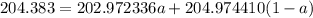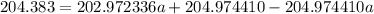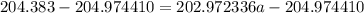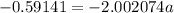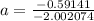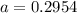Chemistry, 12.07.2019 12:00 tsedeneyaalemu2924
The atomic masses of 203tl and 205tl are 202.972336 and 204.974410 amu, respectively. the average atomic mass of thallium is 204.383 amu. calculate the natural abundances of these two isotopes.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2
You know the right answer?
The atomic masses of 203tl and 205tl are 202.972336 and 204.974410 amu, respectively. the average at...
Questions

Mathematics, 06.05.2020 00:14

Mathematics, 06.05.2020 00:14

English, 06.05.2020 00:14



History, 06.05.2020 00:14


Mathematics, 06.05.2020 00:14

Physics, 06.05.2020 00:14


Mathematics, 06.05.2020 00:14


Mathematics, 06.05.2020 00:14


History, 06.05.2020 00:14

Mathematics, 06.05.2020 00:14



History, 06.05.2020 00:14

History, 06.05.2020 00:14