
Chemistry, 12.07.2019 18:00 darkghostmist
The unknowns are a mixture of ferrous ammonium sulfate and ammonium sulfate. calculate the amount of ferrous ammonium sulfate and the amount of ammonium sulfate needed to prepare a sample that is 10% fe with a total mass of 0.2 g

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
The unknowns are a mixture of ferrous ammonium sulfate and ammonium sulfate. calculate the amount of...
Questions

Computers and Technology, 01.02.2021 20:40








Mathematics, 01.02.2021 20:40





Biology, 01.02.2021 20:40





History, 01.02.2021 20:40

Arts, 01.02.2021 20:40

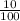 x0.2
x0.2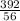 x 0.02
x 0.02

