
Let us assume that fe(oh)2(s) is completely insoluble, which signifies that the precipitation reaction with naoh(aq) (presented in the transition) would go to completion. fe2+(aq)+2naoh(aq) → fe(oh)2(s)+2na+(aq) if you had a 0.500 l solution containing 0.0230 m of fe2+(aq), and you wished to add enough 1.29 m naoh(aq) to precipitate all of the metal, what is the minimum amount of the naoh(aq) solution you would need to add? assume that the naoh(aq) solution is the only source of oh−(aq) for the precipitation.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
Let us assume that fe(oh)2(s) is completely insoluble, which signifies that the precipitation reacti...
Questions

Mathematics, 25.08.2021 20:10

Mathematics, 25.08.2021 20:20


Biology, 25.08.2021 20:20


Mathematics, 25.08.2021 20:20

History, 25.08.2021 20:20


Mathematics, 25.08.2021 20:20

Mathematics, 25.08.2021 20:20


Mathematics, 25.08.2021 20:20

Mathematics, 25.08.2021 20:20


History, 25.08.2021 20:20




Computers and Technology, 25.08.2021 20:20


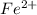 is as follows:-
is as follows:-![Fe^{2+} = 500 mL Fe^{2+} * \frac{0.0230mole\ Fe^{2+}]}{[1 mol\ Fe^{2+}}](/tpl/images/0082/8691/681c2.png) = 11.50 mmol Fe^(2+)
= 11.50 mmol Fe^(2+)![11.50\ mmol\ Fe^{2+} * \frac{[2 \ mol\ NaOH]}{[1 \mol Fe^{2+}}](/tpl/images/0082/8691/9368e.png) = 23.00 mmol NaOH
= 23.00 mmol NaOH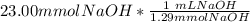 = 17.8 mL NaOH.
= 17.8 mL NaOH.

