
Chemistry, 14.07.2019 04:00 gilbert325
If 3.5 moles of nitrogen monoxide (no) react with 6.0 moles of oxygen gas (o2), how many moles of the product can be formed and how many moles of the excess reactant will be left over when the reaction is complete? show all of your work. unbalanced equation: no + o2 “yields”/ no2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2


Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

You know the right answer?
If 3.5 moles of nitrogen monoxide (no) react with 6.0 moles of oxygen gas (o2), how many moles of th...
Questions




Mathematics, 21.04.2021 21:40

Spanish, 21.04.2021 21:40


Mathematics, 21.04.2021 21:40


Mathematics, 21.04.2021 21:40





Chemistry, 21.04.2021 21:40

English, 21.04.2021 21:40




Mathematics, 21.04.2021 21:40

Mathematics, 21.04.2021 21:40

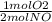 x 3.5 mol NO
x 3.5 mol NO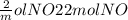 x 3.5 mol NO
x 3.5 mol NO

