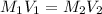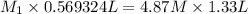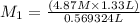Chemistry, 14.07.2019 07:30 prettygirlgwen24
A569324 ml nacl solution is diluted to a volume of 1.33 l and a concentration of 4.87 m. what was the initial concentration? give the molar concentration rounded to the hundredths place without units.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1
You know the right answer?
A569324 ml nacl solution is diluted to a volume of 1.33 l and a concentration of 4.87 m. what was th...
Questions




History, 10.07.2019 12:00

Mathematics, 10.07.2019 12:00




Mathematics, 10.07.2019 12:00

History, 10.07.2019 12:00

History, 10.07.2019 12:00

Social Studies, 10.07.2019 12:00

History, 10.07.2019 12:00


History, 10.07.2019 12:00


Biology, 10.07.2019 12:00


History, 10.07.2019 12:00

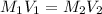
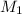 = Initial concentration
= Initial concentration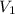 = Initial volume
= Initial volume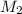 = Final concentration
= Final concentration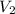 = Final volume
= Final volume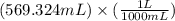
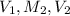 , in dilution equation and will calculate the value of
, in dilution equation and will calculate the value of