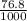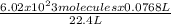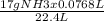Chemistry, 14.07.2019 11:30 Arellano2373
Acommercial refrigeration unit accidentally releases 7.68x10^1 ml of ammonia gas at satp. determine the mass and number of ammonia molecules released.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2
You know the right answer?
Acommercial refrigeration unit accidentally releases 7.68x10^1 ml of ammonia gas at satp. determine...
Questions


Biology, 04.10.2019 23:00


Biology, 04.10.2019 23:00

Health, 04.10.2019 23:00



History, 04.10.2019 23:00




Social Studies, 04.10.2019 23:00

Mathematics, 04.10.2019 23:00

Physics, 04.10.2019 23:00



Health, 04.10.2019 23:00

Spanish, 04.10.2019 23:00

History, 04.10.2019 23:00