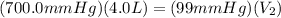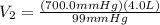Aballoon was partially filled with helium gas at room temperature. it occupied 4.0 liters of volume at 700.0 mmhg atmospheric pressure. when the balloon was released, it traveled upward until it burst at 99 mmhg atmospheric pressure. (neglect any force exerted to stretch the rubber balloon.) what was the volume of the balloon when it burst? l

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2

Chemistry, 23.06.2019 01:30
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
You know the right answer?
Aballoon was partially filled with helium gas at room temperature. it occupied 4.0 liters of volume...
Questions

Mathematics, 29.05.2020 07:01

History, 29.05.2020 07:01





Biology, 29.05.2020 07:01


Mathematics, 29.05.2020 07:01



Mathematics, 29.05.2020 07:01

Mathematics, 29.05.2020 07:01

English, 29.05.2020 07:01



Mathematics, 29.05.2020 07:01

History, 29.05.2020 07:01

Mathematics, 29.05.2020 07:01


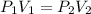
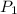 is the initial pressure and
is the initial pressure and 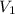 is initial volume.
is initial volume.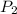 is final pressure and
is final pressure and 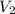 is final volume.
is final volume.