
Chemistry, 15.07.2019 15:30 kikidsgn123
It takes 238./kjmol to break a carbon-iodine single bond. calculate the maximum wavelength of light for which a carbon-iodine single bond could be broken by absorbing a single photon. round your answer to 3 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 23.06.2019 02:00
The bone of a dinosaur and the imprint of a leaf are examples of which kind of fossils? a) index b) body c) amber d) trace
Answers: 1

Chemistry, 23.06.2019 07:30
Assignment directions: pick one of the following chemists and perform a bit of research on him/her. answer the following questions. alice hamilton rosalind franklin marie curie gertrude b. elion ada yonath henry cavendish robert boyle antoine lavoisier mario j. molina svante arrhenius
Answers: 1
You know the right answer?
It takes 238./kjmol to break a carbon-iodine single bond. calculate the maximum wavelength of light...
Questions

History, 05.06.2020 18:58


Mathematics, 05.06.2020 18:58


Chemistry, 05.06.2020 18:58

Computers and Technology, 05.06.2020 18:58




Mathematics, 05.06.2020 18:58




Mathematics, 05.06.2020 18:58



Social Studies, 05.06.2020 18:58


Mathematics, 05.06.2020 18:58

Mathematics, 05.06.2020 18:58

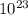 ) = 3.953
) = 3.953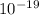 J
J 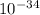 Js X 2.9979
Js X 2.9979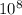 m/s) / (3.953
m/s) / (3.953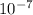 m
m
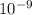 m
m


