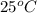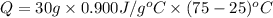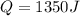Chemistry, 17.07.2019 05:00 rlumanlan549
The specific heat of aluminum is 0.900 j/g•°c. how much heat is required to raise the temperature of a 30.0 g block of aluminum from 25.0°c to 75.0°c?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
You know the right answer?
The specific heat of aluminum is 0.900 j/g•°c. how much heat is required to raise the temperature of...
Questions

Biology, 13.07.2019 01:30


Mathematics, 13.07.2019 01:30








Mathematics, 13.07.2019 01:30

Mathematics, 13.07.2019 01:30



Mathematics, 13.07.2019 01:30





Mathematics, 13.07.2019 01:30

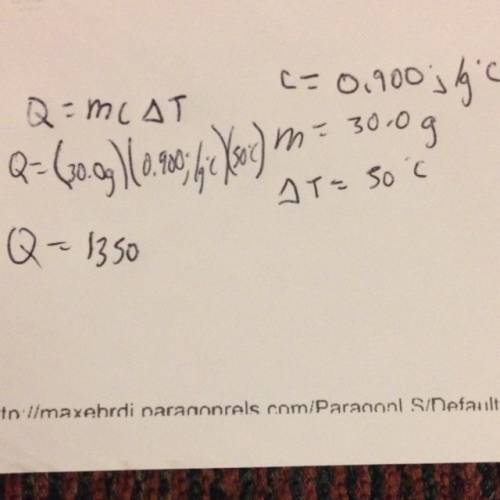
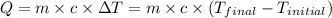
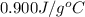
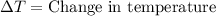
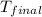 = final temperature =
= final temperature = 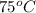
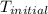 = initial temperature =
= initial temperature =