Chemistry, 17.07.2019 08:00 crawford184232323234
What is the value of the equilibrium constant for this redox reaction? 2ag+(aq) + zn (s) → 2ag (s) + zn2+(aq) e = +1.56 v k=10(ne∘0.0592) k = 2.25 × 10-26 k = 2.25 × 1026 k = 5.04 × 10-52 k = 5.04 × 1052

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2


Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1
You know the right answer?
What is the value of the equilibrium constant for this redox reaction? 2ag+(aq) + zn (s) → 2ag (s)...
Questions

Mathematics, 19.12.2019 03:31





Mathematics, 19.12.2019 03:31


Mathematics, 19.12.2019 03:31


Mathematics, 19.12.2019 03:31


Biology, 19.12.2019 03:31


Mathematics, 19.12.2019 03:31



Mathematics, 19.12.2019 03:31

Mathematics, 19.12.2019 03:31

Mathematics, 19.12.2019 03:31

Health, 19.12.2019 03:31

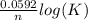 ,
,