
Chemistry, 18.07.2019 13:00 AkramMasoud
5fe2+ + mno4- + 8h+ yields 5fe3+ + mn2+ + 4h2o in the equation above, a: h+ ions are reduced b: fe2+ ions are oxidized c: h+ is the oxidizing agent d: mno4- is the reducing agent

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

You know the right answer?
5fe2+ + mno4- + 8h+ yields 5fe3+ + mn2+ + 4h2o in the equation above, a: h+ ions are reduced b: f...
Questions


Mathematics, 13.11.2020 16:40


Chemistry, 13.11.2020 16:40



English, 13.11.2020 16:40

Mathematics, 13.11.2020 16:40

English, 13.11.2020 16:40





Mathematics, 13.11.2020 16:40






Mathematics, 13.11.2020 16:40

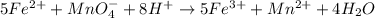
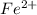 changes to
changes to 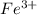 , that is, it is oxidizing.
, that is, it is oxidizing.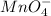 changes from
changes from 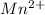 , that is, changes from +7 to +2.
, that is, changes from +7 to +2. 

