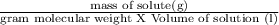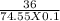Chemistry, 20.07.2019 14:30 familyk0jj3
Potassium chloride (kcl) is sometimes used to treat low blood potassium levels. calculate the concentration of a saturated solution of potassium chloride at 20°c. question continued-? hint: the formula for molarity, a common concentration unit, is m = moles/volume of solution expressed in units of mol/l. you will need to convert from grams of kcl to moles. assume the volume of solution is 100 ml. show your work! info: 36g of kcl is the max 100ml of water could hold at 20°c.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2

Chemistry, 23.06.2019 09:30
If the solubility of a gas in water is 1.22g/2.75 atm, what is it’s solubility (in g/l) at 1.0 atm
Answers: 1

Chemistry, 23.06.2019 10:30
Can anyone explain 1. review your spectrometry data and use the known elements to identify the star's composition. which unknown elements make up this star? justify your element selections. 2. in parts i and ii of the lab, what happened to the electrons of each element to produce the different colors of light? explain your answers using important terms from the lesson and information provided in the laboratory. 3. stars composed of heavier (more massive) elements are often slightly older than stars made predominantly from hydrogen and helium. based on your data, is the newly discovered star a younger star? explain your answer.
Answers: 2
You know the right answer?
Potassium chloride (kcl) is sometimes used to treat low blood potassium levels. calculate the concen...
Questions


Mathematics, 29.10.2020 22:20

Mathematics, 29.10.2020 22:20


Arts, 29.10.2020 22:20

History, 29.10.2020 22:20

Spanish, 29.10.2020 22:20

Biology, 29.10.2020 22:20

Mathematics, 29.10.2020 22:20


History, 29.10.2020 22:20


Mathematics, 29.10.2020 22:20

Health, 29.10.2020 22:20

History, 29.10.2020 22:20

Computers and Technology, 29.10.2020 22:20

English, 29.10.2020 22:20

English, 29.10.2020 22:20


Arts, 29.10.2020 22:20