Many exergonic reactions fail to happen at a reasonable rate (e. g. conversion of diamonds to charcoal). This is due to the fact that their activation energy may be too high to overcome. Which of the following correctly describes the reason for this?
A. The free energy of the transition state is much lower than the free energy of the reactants.
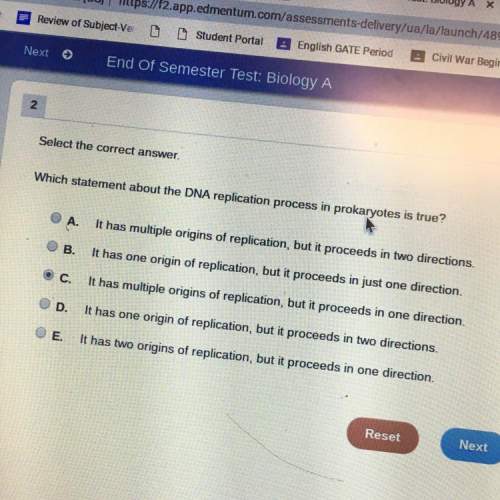
B. The free energy of the transition state is much higher than the free energy of the reactants.
C. The enzyme that catalyzes the reaction needs time to interact chemically with the substrate(s).
D. The interaction between the enzyme and substrate(s) involve a decrease in entropy, which can’t happen without an input of energy.

Answers: 1
Another question on Biology

Biology, 22.06.2019 01:00
If water were a non polar molecule, how would its properties be different ? a.) water would be a much lighter molecule b.) water would be able to climb up inside plants c.)water would not be able to form hydrogen bonds d.) water would stick together much more strongly
Answers: 2


Biology, 22.06.2019 04:50
What part of the brain is highlighted in the diagram below?
Answers: 2

Biology, 22.06.2019 08:00
In a vegetable garden 45 bean plants have been eaten by deer 5 are untouched what is the frequency of bean plants in the garden that have been eaten?
Answers: 3
You know the right answer?
Many exergonic reactions fail to happen at a reasonable rate (e. g. conversion of diamonds to charco...
Questions


Mathematics, 09.09.2021 05:50


Mathematics, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50

History, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50



Mathematics, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50


Mathematics, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50


Biology, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50

English, 09.09.2021 05:50

Mathematics, 09.09.2021 06:00

Mathematics, 09.09.2021 06:00